Following the news from the United Kingdom
Provided by AGPAI-Based Clinical Trials Solution Providers Market Grows at 22.1% CAGR Through 2033 | Persistence Market Research
AI-based clinical trial solutions enhance patient recruitment, data management, and trial efficiency, enabling faster, accurate, and evidence-driven research.
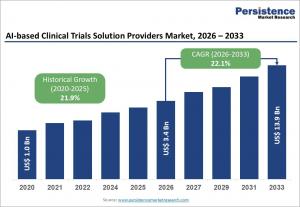
LONDON, UNITED KINGDOM, May 5, 2026 /EINPresswire.com/ -- The global AI-based clinical trials solution providers market is poised for significant expansion, with its valuation expected to reach US$13.9 billion by 2033, up from an estimated US$3.4 billion in 2026. This represents a robust compound annual growth rate (CAGR) of 22.1% during the forecast period, underscoring the growing urgency among pharmaceutical and biotechnology companies to modernize conventional clinical development processes. Traditional clinical trials are often hampered by long timelines, high failure rates, and operational inefficiencies, prompting stakeholders to adopt AI-driven solutions that optimize every stage of the trial lifecycle.
Download Your Free Sample & Explore Key Insights: https://www.persistencemarketresearch.com/samples/32855
AI Transforming Clinical Trial Operations
Artificial intelligence has emerged as a transformative force in clinical research, enhancing protocol design, patient recruitment, site selection, data management, and real-time monitoring. Unlike replacing regulatory frameworks, AI complements existing processes by increasing accuracy, reducing manual workloads, and enabling faster, evidence-based decisions. The surge in complex trials, particularly in precision medicine, rare diseases, and targeted therapies, has further accelerated the adoption of AI-driven clinical trial solutions. By leveraging AI, sponsors can manage intricate trial designs and large datasets more efficiently, improving outcomes and minimizing risks associated with conventional methods.
Regional Insights
North America is expected to dominate the market in 2026, holding approximately 40% of the share. This leadership is driven by strong innovation ecosystems, advanced digital infrastructure, and supportive regulatory frameworks. The U.S. FDA’s increasing recognition of AI/ML tools has spurred widespread adoption among pharmaceutical companies and contract research organizations (CROs). Europe is also emerging as a key market, with initiatives such as Horizon Europe and the European Medicines Agency fostering AI integration through funding, standardization, and collaborative innovation. Asia Pacific is anticipated to be the fastest-growing region, buoyed by government support, expanding healthcare digitization, and large, diverse patient populations that facilitate scalable AI training and clinical trial execution.
Therapeutic Areas and Trial Phases
Oncological disorders are projected to lead revenue in the AI-based clinical trials market, accounting for roughly 40% of the share in 2026. The high volume of oncology trials, along with biomarker-driven and precision medicine approaches, makes oncology highly suitable for AI application. Cardiovascular trials are expected to witness the fastest growth, driven by rising disease prevalence and the integration of wearable devices and remote monitoring to enhance real-time patient data analysis.
Phase III trials are predicted to dominate the market, representing over 50% of revenue in 2026 due to their complexity, large patient populations, and significant regulatory stakes. AI solutions optimize site selection, predict enrollment timelines, and enable risk-based monitoring, thereby improving efficiency. Meanwhile, Phase I trials are expected to grow rapidly as AI facilitates early-stage drug development through adaptive protocols, biomarker-based patient selection, and toxicity prediction, reducing attrition rates and accelerating go/no-go decisions.
Get Custom Insights Designed for Your Businecss: https://www.persistencemarketresearch.com/request-customization/32855
Market Drivers
Technological advancements in AI and machine learning, coupled with integration with real-world evidence (RWE), are key growth drivers. AI enables predictive modeling, automated data processing, and intelligent protocol design by analyzing electronic health records, genomics, and imaging data. The use of RWE provides continuous, real-time insights, improving trial adaptability, patient recruitment, and retention. Additionally, the rising prevalence of chronic diseases, including cancer, cardiovascular conditions, and neurological disorders, is creating demand for complex, large-scale trials where AI can enhance protocol design, patient stratification, and outcome prediction.
Market Restraints
Despite its growth potential, the market faces challenges such as data privacy, security concerns, and algorithmic bias. Handling sensitive patient data requires strict compliance with regulatory standards while maintaining accessibility for AI systems. Algorithmic bias, resulting from unrepresentative training datasets, can affect patient selection and trial outcomes. Interoperability issues with legacy systems and resistance to organizational change further complicate AI adoption, necessitating investments in infrastructure modernization and stakeholder training.
Opportunities
AI integration with decentralized and hybrid trial models represents a major opportunity. Remote monitoring, wearable devices, and telemedicine combined with AI enable real-time data analysis, enhance patient engagement, and reduce operational costs. AI-driven bias mitigation and diversity tools are increasingly adopted to ensure representative patient populations, improving trial reliability, regulatory compliance, and trust in clinical research outcomes.
Checkout Now & Download Complete Market Report: https://www.persistencemarketresearch.com/checkout/32855
Market Segmentation
By Therapeutic Areas
Cardiovascular Disorders
CNS Disorders
Infectious Disorders
Metabolic Disorders
Oncological Disorders
By Trial Phase
Phase I
Phase II
Phase III
By Application
Clinical Trial Design
Patient Enrichment and enrollment
Investigator and site selection
Patient Monitoring
Medication Adherence
By Region
North America
Europe
East Asia
South Asia & Oceania
Latin America
Middle East & Africa
Competitive Landscape
The market exhibits moderate fragmentation, with both established technology firms and niche AI startups driving innovation. Key players include Unlearn.ai, Saama, Antidote Technologies, Deep6.ai, Innoplexus, Mendel Health, Median Technologies, SymphonyAI, and BioAge Labs. Companies are leveraging machine learning, natural language processing, and predictive analytics to optimize clinical operations, reduce timelines, and improve patient outcomes. Strategic collaborations, platform expansion, and product innovation are shaping the competitive dynamics in this rapidly evolving market.
Recent Developments
In December 2025, Mass General Brigham launched AIwithCare, a platform using generative AI to enhance clinical trial screening and patient recruitment. In July 2025, PhaseV introduced the ClinOps AI platform, designed to optimize site selection and overall trial efficiency through predictive analytics. These developments highlight the growing emphasis on AI-driven solutions to streamline clinical trials and accelerate drug development.
The AI-based clinical trials solution providers market is set for transformative growth, driven by technological innovation, rising clinical complexity, and a global push toward data-driven, patient-centric research approaches.
Read Related reports:
Arthroscopic Devices Market: The global arthroscopic devices market grows from US$ 8.6 Bn in 2026 to US$ 12.1 Bn by 2033, at a 4.9% CAGR during forecast 2026–2033.
Biologics Safety Testing Market: The global biologics safety testing market grows from US$ 4.6 Bn in 2026 to US$ 8.8 Bn by 2033, at a 9.7% CAGR during forecast 2026–2033.
Persistence Market Research
Persistence Market Research Pvt Ltd
+1 646-878-6329
email us here
Visit us on social media:
LinkedIn
Instagram
Facebook
YouTube
X
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.